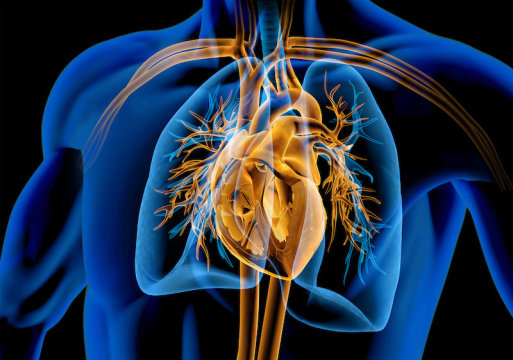
Because the heart, unlike other organs, cannot heal itself after injury, heart disease—the top cause of mortality in the U.S.—is particularly lethal. For this reason, tissue engineering will be crucial for the development of cardiac medicine, ultimately leading to the mass production of a whole human heart for transplant.
Researchers need to duplicate the distinctive structures that make up the heart in order to construct a human heart from the ground up. This involves re-creating helical geometries, which cause the heart to beat in a twisting pattern. It has long been hypothesized that this twisting action is essential for pumping blood at high rates, but establishing this has proven problematic, in part because designing hearts with various geometries and alignments has proven difficult.
The first biohybrid model of human ventricles with beating cardiac cells that are helically aligned has now been created by bioengineers from the Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS), and it has demonstrated that muscle alignment does, in fact, significantly increase the amount of blood the ventricle can pump with each contraction.
Focused Rotary Jet Spinning (FRJS), a novel additive textile manufacturing technique, allowed for the high-throughput manufacture of helically aligned fibers with diameters ranging from several micrometers to hundreds of nanometers, which enabled this development. FRJS fibers, which direct cell alignment, were created at SEAS by Kit Parker’s Disease Biophysics Group, enabling the development of controlled tissue engineered structures.
The Tarr Family Professor of Bioengineering and Applied Physics at SEAS and the paper’s senior author, Parker, said that the research represents a significant advance in the field of organ biofabrication and moves us one step closer to our ultimate objective of creating a human heart for transplant.
This piece’s inspiration came from a long-solved mystery. In his important book Tractatus de Corde, English surgeon Richard Lower, who listed John Locke among his associates and King Charles II among his patients, first described the spiral-like arrangement of cardiac muscles in 1669.
Over the ensuing three centuries, medical professionals and researchers developed a more thorough grasp of the anatomy of the heart, but the function of those spinning muscles has remained frustratingly challenging to research.
Edward Sallin, a former chair of the Department of Biomathematics at the University of Alabama Birmingham Medical School, asserted in 1969 that a substantial ejection fraction—the proportion of blood that the ventricle pumps with each contraction—requires a helical alignment of the heart.
John Zimmerman, a postdoctoral scholar at SEAS and co-first author of the publication, said that the team’s objective was to create a model that would allow them to test Sallin’s theory and investigate the relative significance of the heart’s helical configuration.
The SEAS researchers employed the FRJS system to regulate the alignment of spun fibers on which they could grow cardiac cells in order to test Sallin’s idea.
A liquid polymer solution is poured into a reservoir and forced out via a small aperture by centrifugal force while the device spins in the first stage of FRJS, which operates similarly to a cotton candy machine. The solvent evaporates as the solution exits the reservoir, and the polymers solidify to form fibers. When the fibers are deposited on a collector, a concentrated airstream regulates their orientation. The group discovered that by tilting and rotating the collector, the stream’s fibers would line up and cling to it as it spun, like the helical shape of heart muscles.
Huibin Chang, a postdoctoral scholar at SEAS and co-first author of the work, stated that the human heart “really comprises numerous layers of helically aligned muscles with varied degrees of alignment.” With FRJS, we are able to precisely reproduce these intricate structures, creating single and even four chambered ventricle architectures.
FRJS can swiftly spin fibers at the single micron scale, or around fifty times smaller than a single human hair, in contrast to 3D printing, which becomes slower as features increase smaller. When creating a heart from scratch, this is critical. Consider the extracellular matrix protein collagen, which is likewise one micron in diameter and is found in the heart. To 3D print the whole amount of collagen in the human heart at this resolution would take more than 100 years. In a single day, FRJS can complete it.
The ventricles were spun before being implanted with rat or human stem cell derived cardiomyocyte cells. The scaffold was covered in many thin layers of beating tissue after approximately a week, with the cells aligning themselves according to the fibers underneath.
Researchers examined ventricles created from helical aligned fibers versus those made from circumferentially aligned fibers in terms of ventricle deformation, electrical signaling speed, and ejection %. They discovered that the helically aligned tissue performed better than the circumferentially aligned tissue on every front.
Since 2003, Parker’s team has focused on figuring out how the links between the heart’s structure and function are affected by illness in pathological ways. “In this instance, we returned to a previously untested finding on the helical structure of the heart’s laminar architecture. We were fortunate to be able to construct a new manufacturing platform that allowed us to verify Professor Sallin’s theoretical prediction from more than 50 years ago and answer this age-old query.”
The scientists also showed that the procedure could be expanded up to the size of a real human heart and even larger, to that of a Minke whale heart (they didn’t seed the larger models with cells because it would require billions of cardiomyocyte cells).
The John A. Paulson School of Engineering and Applied Sciences at Harvard contributed the materials.